Governance
BBCIC Governance
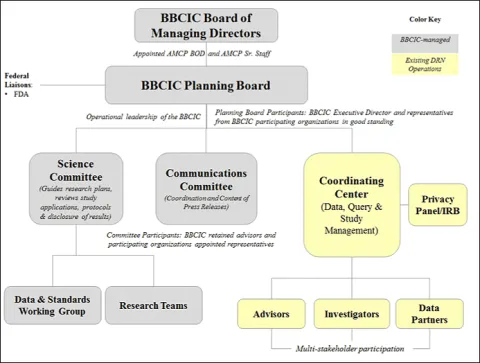
The BBCIC is a participant driven consortium governed by a Board of Managing Directors, a Planning Board and a Science Committee.
Board of Managing Directors
The BBCIC Board of Managing Directors is responsible for oversight of the AMCP BBCIC and consists of the following positions:
- Babette S. Edgar, BS Pharmacy, MBA, RPh, AMCP President
- William Francis, BSPharm, MBA, RPh, AMCP Treasurer
- Susan A. Cantrell, RPh, CAE, AMCP CEO
- At Large Directors:
- Bradley C Martin, PharmD, PhD, Professor and Head Division of Pharmaceutical Evaluation and Policy, University of Arkansas for Medical Sciences College of Pharmacy
- Philip Gerbino, PharmD, President Emeritus, University of the Sciences and Past President, American Pharmacist Association
Planning Board
The Planning Board is responsible for the BBCIC charter, operational policies & procedures and research plans. The Planning Board also oversees public communications regarding AMCP BBCIC activities and findings. Click Here to view the members of the Planning Board.
Science Committee
The Science Committee is responsible for the development and conduct of the overall research plan and publication plans. The Science Committee consists of scientific experts from the participating organizations. Click Here to view the members of the Science Committee.
Research Teams
The Research Teams reflect the various BBCIC Participants and include data, clinical, epidemiology, statistics and HEOR expertise. The BBCIC Science Committee appoints additional subject matter experts where the expertise is not available from our Participant organizations. Research Teams write formal Research Protocols to be approved by the Science Committee and work with the Coordinating Center to execute the completion and publication of the protocol results. Click Here to view the members of the Research Teams.
BBCIC Leadership
Cate Lockhart, PharmD, PhD
Executive Director, Biologics and Biosimilars Collective Intelligence Consortium (BBCIC)
675 North Washington Street, Suite 220, Alexandria, VA 22314
@email
703-684-2646
BBCIC Charter
The BBCIC Charter was approved by the BBCIC Board of Managing Directors on October 23, 2015. The BBCIC Charter details the principles and policies governing all BBCIC research projects.
Click Here to access the BBCIC Charter PDF.
BBCIC Antitrust Policy
The BBCIC follows strict antitrust guidelines. BBCIC policy is to comply fully and strictly with all federal and state antitrust laws.
Click Here to access the BBCIC Charter PDF.
BBCIC Transparency of Its Science-Driven Approach
The BBCIC focus is its transparent science-driven research. The BBCIC scope does not include advocacy or political functions, including taking a position or commenting on legislative or regulatory proposals. The AMCP BBCIC is committed to transparency in all aspects of its research and operations.
The BBCIC requires all BBCIC Research Reports to be communicated promptly in an appropriate scientific forum. The Research Team will register all BBCIC Research Protocols in ClinicalTrials.gov prior to initiating the study. Methodological and other projects will be registered at HSRProj
- The BBCIC requires that BBCIC Research Reports be submitted for publication within 6 months after the completion of the Research Report. When published, the publication will be posted on the BBCIC.org site.
- Where appropriate, the Research Team will report verified findings of interest to regulatory authorities (see Policy 2.14 Safety-related Findings of Interest)
- This page will be the future location for the details of all BBCIC Research Protocols